 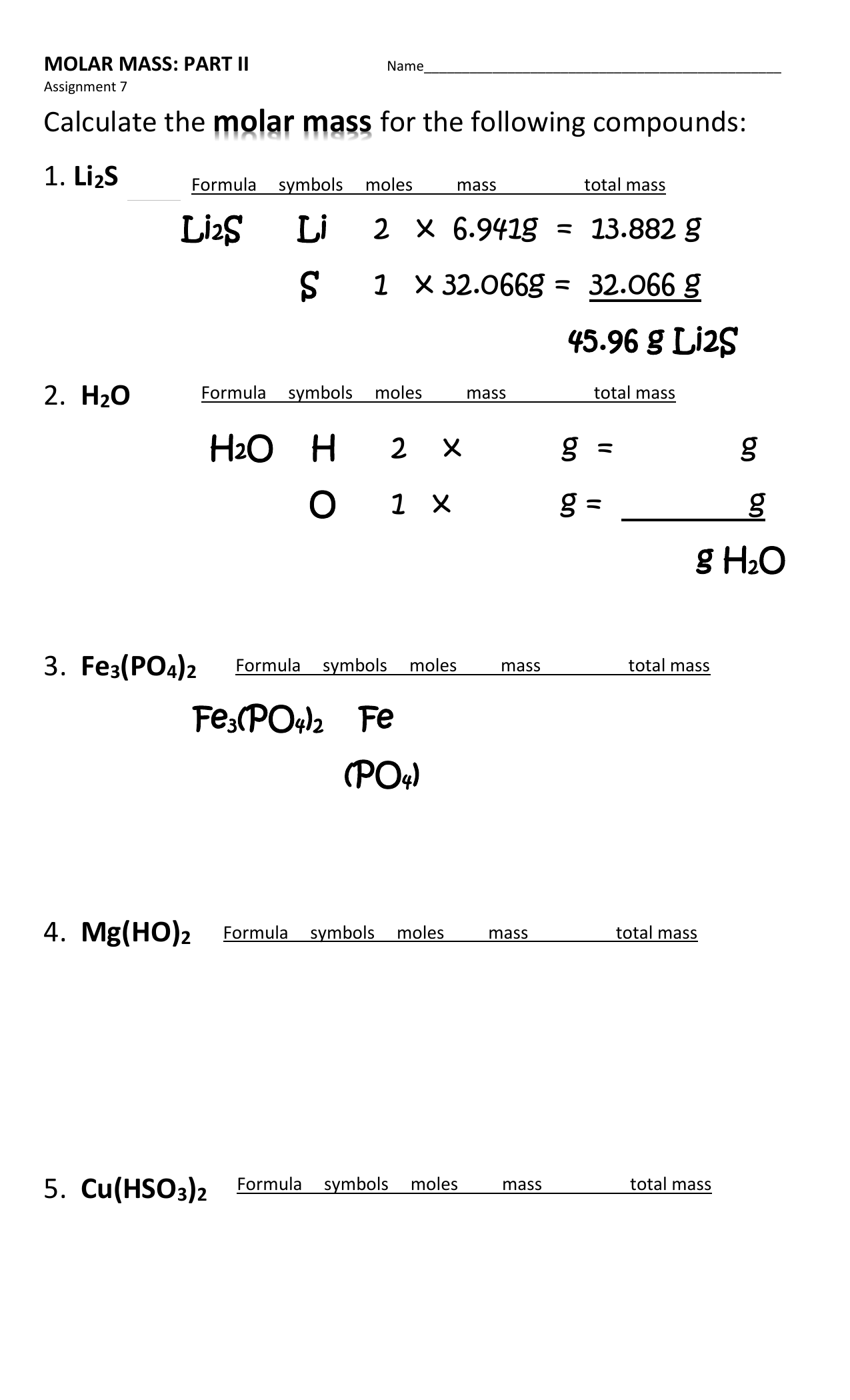
Most of the time, molecular mass and molar mass are interchangeable. This is because the calculation uses the average atomic masses from the periodic table, which are weighted mass averages based on isotope abundance of natural elements. Technically, the usual molecular mass calculation is really a molar mass calculation. So, while you report molecular mass in daltons or amu, molar mass is in kilograms per mole (kg/mol) or grams per mole (g/mol). The molar mass of a compound is the sample mass divided by the number of moles. Difference Between Molecular Mass and Molar Mass The molecular mass of sulfuric acid is equal to 98.078 amu or 98.078 grams/mole. Molecular mass of H 2SO 4 = (2 x mass of hydrogen) (1 x mass of sulfur) (4 x mass of oxygen) What is the molecular mass of sulfuric acid (H 2SO 4)?Įach sulfuric acid molecule contains 2 hydrogen atoms, 1 sulfur atom, and 4 oxygen atoms. This means one mole of water weighs 18.015 grams. This is how you can find the molecular weight of a molecule. Remember that one amu is equivalent to 1 gram/mole. The molecular mass of water is 18.015 amu. Molecular mass of H 2O = 2.016 amu 15.999 amu The molecular mass of water will be 2 masses of hydrogen plus the mass of oxygen. The periodic table shows the atomic mass of hydrogen is 1.0008 amu and the atomic mass of oxygen is 15.999 amu. This means one water molecule contains two hydrogen atoms and one oxygen atom. If there is no subscript following an element symbol, it means there is one atom of that element in the formula.įor example, water has the molecular formula H 2O. The number of atoms of each element is its subscript in the molecular formula. Once you know the atomic makeup of the molecule, you use the periodic table to find the mass of each atom and add them together. Add up the atomic masses of each element.Ĭalculating molecular mass requires that you know the number and type of atoms that make up the molecule.Multiply each atomic mass by its subscript in the formula.Look up the atomic masses of each element in the formula.Write the molecular formula for the molecule or compound.Here is how to find molecular mass, example calculations, and a look at the difference between molecular mass and molar mass. Usually, you report molecular mass (m) in units of daltons (Da) or atomic mass units (amu or u). Molecular mass is the mass of one molecule. Molecular mass is the mass of a molecule or compound according to its molecular formula. If they are next to each other, multiply them to get the answer, if one is on top of the other, divide the top one by the bottom one to get the answer.This entry was posted on Augby Todd Helmenstine (updated on August 17, 2022) To get the formula from the triangle, cover up the value you want to calculate and use the remaining two values. To remember the formulas in this section, you can use the formula triangles. 
For example, about 98.9% of naturally occurring carbon is 12C ('Carbon 12') and about 1.1% is 13C so carbon has an atomic weight of \begin Formula Triangles 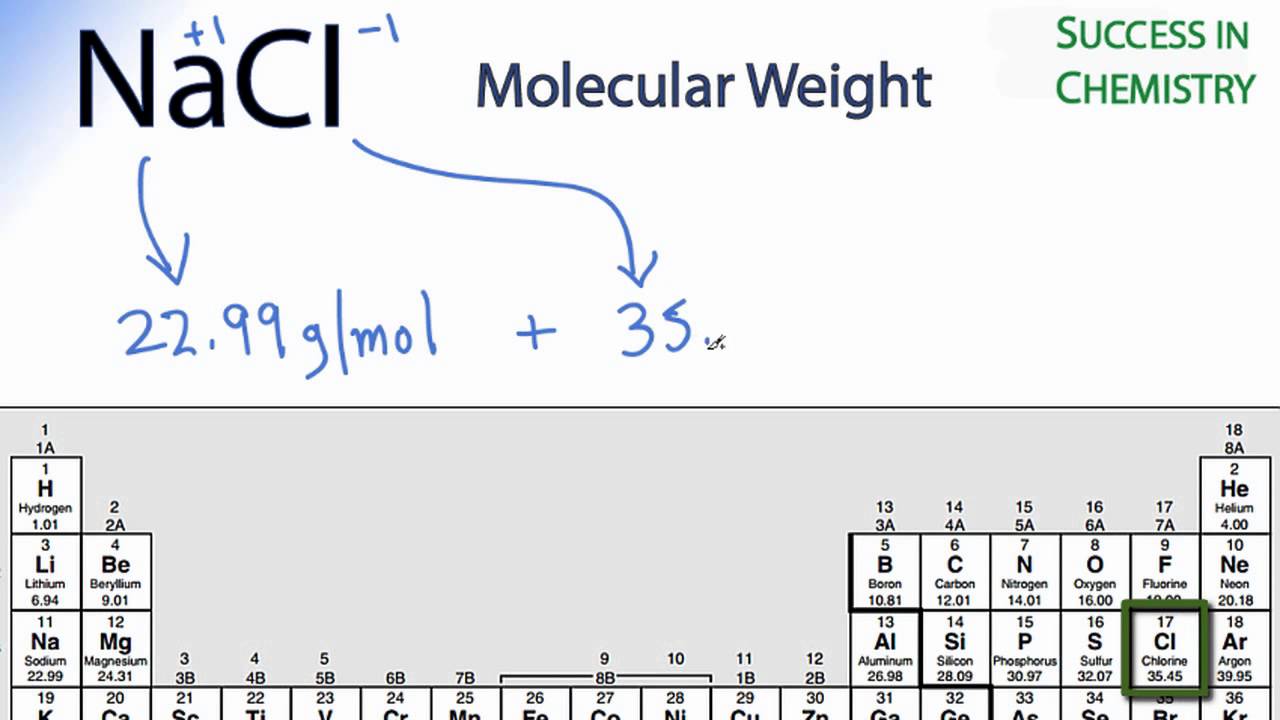 
The atomic weight (or atomic mass) of an element tells us on average how much one atom of a given element weighs, taking into account typical proportions of isotopes. Contents Toggle Main Menu 1 Atomic Weight 2 Molecular Weight 3 Moles 4 Solutions 4.1 1) Molarity 4.2 2) Mass Concentration 4.3 A note on units for concentrations 4.4 Diluting Solutions 5 Test yourself Atomic Weight 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
